 
They don't really have a color, we just use that idea because it's sort of helpful, just like using the idea of positive and negative for charges is helpful. IN the field of quantum chromodynamics, we say that particles have a color. Bohr model of the hydrogen atom was the first atomic model to successfully explain the radiation spectra of atomic hydrogen. It's just the name that we've given a quark that has certain properties. There's nothing unusually strange about the strange quark. There's a charmed quark and a strange quark. THere's nothing "up-ish" about an up quark. "Up" and "down" don't have any meaning other than to identify the type of quark. 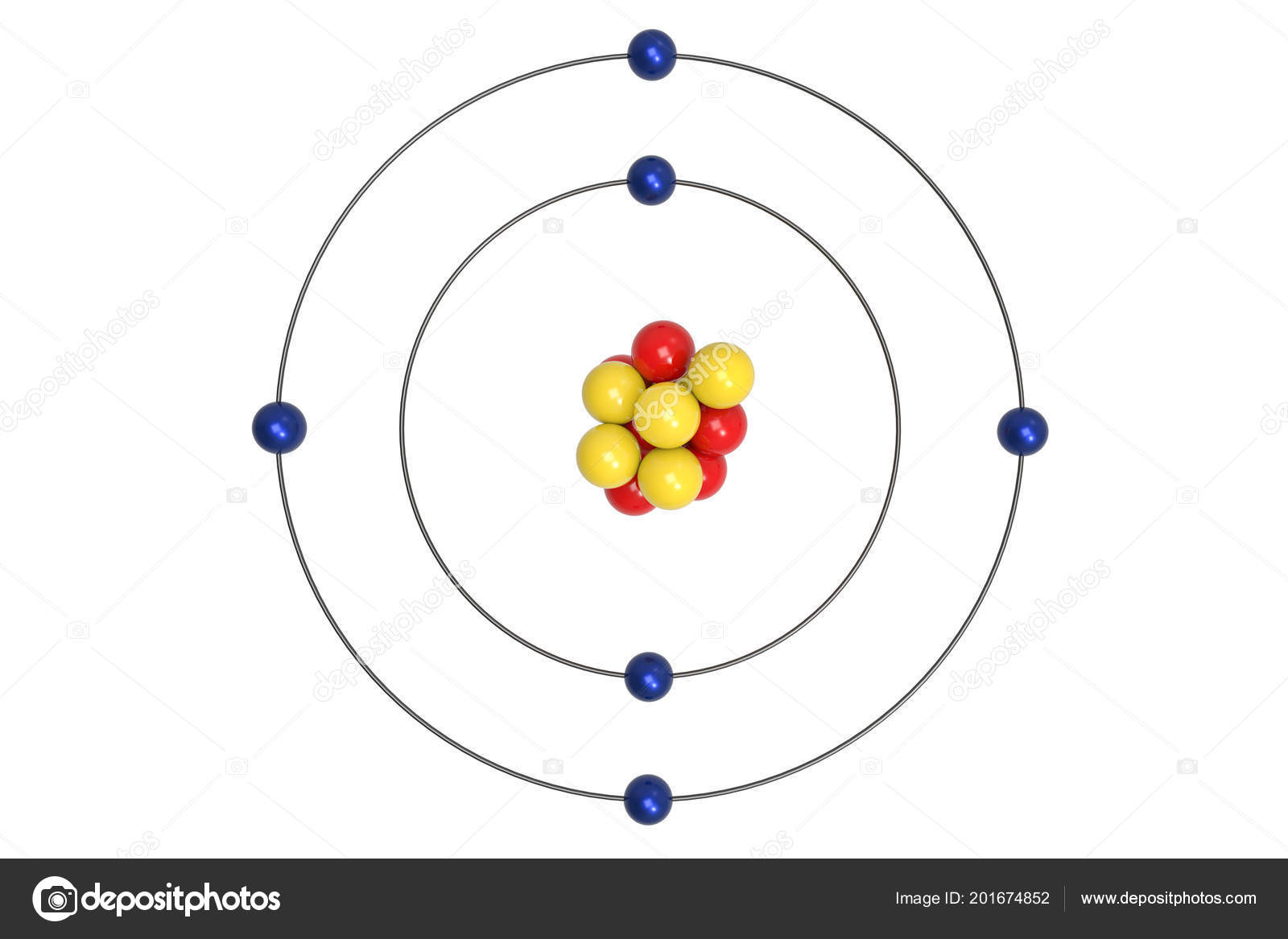
A quark can be an up quark or a down quark. We have other properties like this for other particles. It's just a characteristic that particles have or don't have, and we know what the effect is of having or not having that property. "What is spin" doesn't have any deeper answer than does the question "what is charge". But there is no point in trying to answer "what is electron spin" by referring to some familiar object like a ball or a planet, because electrons are not balls or planets, they are their own thing, and you just have to accept that they have a property whose effects we understand very well, and we happen to have named it spin. Still, they have a property that works sort of like an orbit (orbitals) and they have a property that works sort of like rotation about an axis (spin). Using this concept actually helped a bit, just like Bohr's imagination of little orbits helped a bit, but again, the electrons are not like little planets: they don't revolve around the nucleus, and they don't rotate on their axes. Just as Bohr imagined that the atoms were little planets revolving around the nucleus as though it were a sun, other scientists tried to extend that idea by imagining that the little planets, just like real planets, had spin. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. It got named spin back when people were working with the Bohr model and trying to extend it to atoms beyond hydrogen. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. 
The word ‘atom’ actually comes from Ancient Greek and roughly translates as. In fact, we have to go all the way back to Ancient Greece to find its genesis. Though our graphic starts in the 1800s, the idea of atoms was around long before. Spin is just a property that electrons (and other particles) have. This graphic takes a look at the key models proposed for the atom, and how they changed over time. The figure includes a diagram representing the relative energy levels of the quantum numbers of the hydrogen. For another layer, you can take the fact that you can never cool anything down to exactly 0K (-273.15C) (although you can get close) and so nothing will ever have 0 velocity. Figure 6.2.2: The horizontal lines show the relative energy of orbits in the Bohr model of the hydrogen atom, and the vertical arrows depict the energy of photons absorbed (left) or emitted (right) as electrons move between these orbits. (1 value in a range of reals is like trying to throw a dart at a dartboard with an infinitely thin wire and hitting the wire). With this uncertainty, the velocity is almost definitely not 0. As we can't physically measure to perfect accuracy, there is an uncertainty in both measurements of the degree that we know it's probably stationary and it's probably 'over there'. So, if you know with 0 uncertainty what the velocity is, then you have no idea where it is, and all future involvement of the particle is pretty much irrelevant (how is the electron going to diffract around an atom if the electron is in a different galaxy?). The square (probability function) shows that it has an equal chance of being anywhere. If you take the infinite wavelength interpretation, then it would be nearly 0 (1/inf) but constant everywhere. \), the lines in this series correspond to transitions from higher-energy orbits (n > 2) to the second orbit (n = 2).Isn't to do with the fact that the velocity is not quite 0? if you know it is exactly 0 then the uncertainty in the position is infinite as well (momentum is a function of velocity, so delta P = 0 -> delta V = 0 -> delta X = inf) therefore it has an equal probability of being anywhere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
